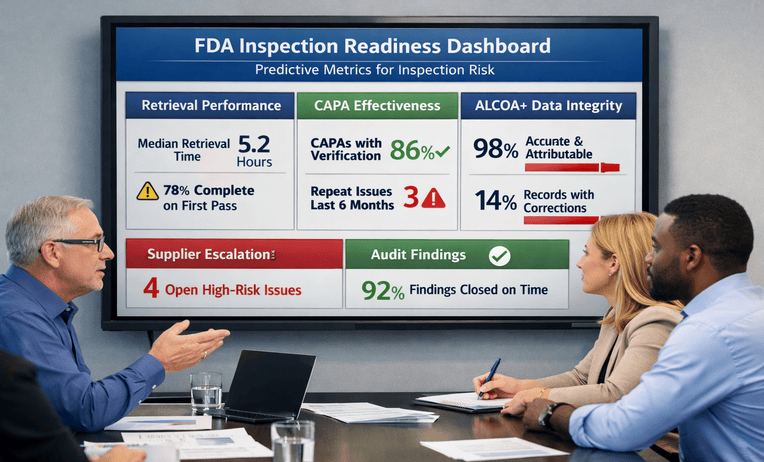
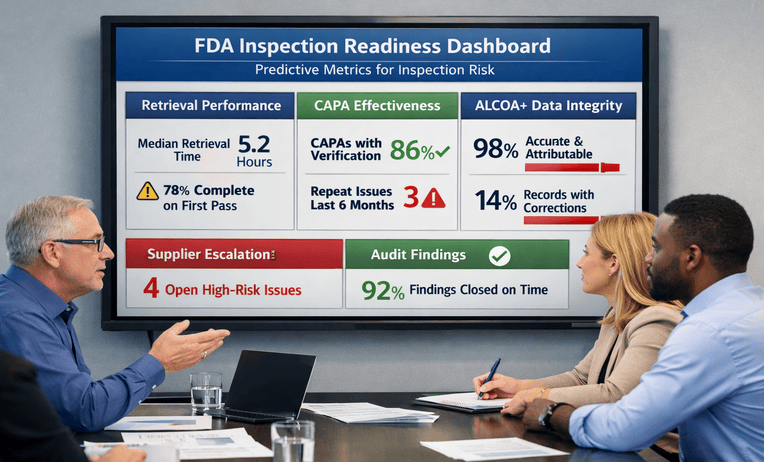
Understanding CLIA: Key Regulations for Lab Safety
Understanding CLIA regulations is essential for maintaining laboratory safety, testing accuracy, and regulatory compliance in clinical diagnostic settings. CLIA standards establish requirements for quality control, personnel training, proficiency testing, and operational procedures to help laboratories deliver reliable patient results while supporting safe and effective healthcare practices.