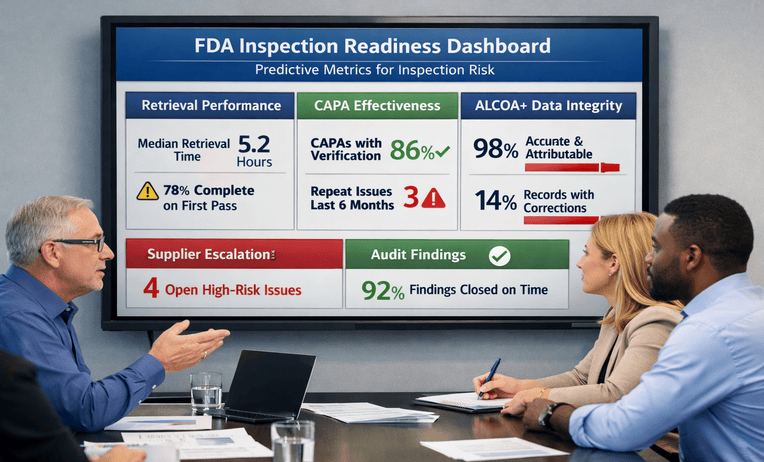
Pharmaceutical inspection readiness is not a one-time project that starts when an inspection notice arrives. For quality, manufacturing, and regulatory leaders, it is the ability to prove that the site operates in control, that records support decisions, and that staff can respond consistently when FDA begins asking detailed questions.
If you are searching for the best pharmaceutical inspection readiness approach, the practical issue is not whether your team has procedures. Instead, the issue is whether deviations, CAPA, batch records, supplier oversight, validation, training, and data integrity controls all hold together under pressure. Therefore, readiness should test both system quality and human execution.
In practice, pharmaceutical inspection risk often appears where the technical story and the operating story start to separate. A batch record may look complete, yet investigation follow-through may be weak. A CAPA may be closed, yet effectiveness may remain unclear. Meanwhile, one function may answer confidently while another gives a different explanation for the same process.
As a result, strong pharmaceutical inspection readiness means building one defensible operating picture. The written procedure, the executed process, the electronic record, and the verbal explanation must align clearly enough to withstand scrutiny.
Quick answer
The best pharmaceutical inspection readiness model is a risk-based review of manufacturing controls, quality system records, data integrity practices, and inspection behavior before FDA arrives. In short, pharmaceutical inspection readiness should help a company retrieve evidence quickly, explain control clearly, and correct weaknesses before they become inspection findings.
What you get
- A focused pharmaceutical inspection readiness assessment
- Review of quality, manufacturing, validation, and supplier oversight controls
- Document retrieval and inspection room practice
- Interview coaching for leaders and subject matter experts
- Data integrity and Part 11 readiness checks where relevant
- A prioritized action tracker for readiness gaps
- Better coordination across quality, operations, validation, and leadership
When you need this
- An FDA inspection is likely in the near term
- The site has grown, changed systems, or expanded operations
- Leadership wants a realistic view of current readiness
- Repeated deviations or weak CAPA closure are creating concern
- Several vendors or CMOs support product quality
- The team needs a clear path from assessment to remediation
Table of contents
- What pharmaceutical inspection readiness should cover
- Scope and deliverables that matter
- Timeline example and required inputs
- Common pharmaceutical inspection readiness gaps
- How BioBoston works in practice
- How to choose the right readiness partner
- Case study
- Next steps
- FAQs
- Why teams use BioBoston Consulting
What pharmaceutical inspection readiness should cover
Pharmaceutical inspection readiness should cover how the site actually operates from a quality and manufacturing standpoint. Importantly, inspectors do not only review isolated records. Instead, they look for whether the overall system shows control across batch execution, investigations, oversight, validation, and decision-making.
Core areas often include batch records, deviations, CAPA, change control, complaints, training, supplier oversight, cleaning and qualification where relevant, process validation, and management review. Therefore, a readiness review should test whether these elements connect into one consistent control model.
For many pharmaceutical companies, data integrity is also central. If electronic systems support manufacturing or quality decisions, readiness should include FDA 21 CFR Part 11, audit trails, access controls, review practices, record retention, and ALCOA+ principles. In practice, weak control over electronic evidence often creates unnecessary inspection pressure.
Meanwhile, broader governance and risk decisions are often evaluated through a lens consistent with ICH Q9 and ICH Q10. However, the practical inspection question remains simple. Can the company show what happened, why it happened, how it was reviewed, and how it was controlled.
You can review BioBoston’s inspection support here: FDA Inspection Readiness. Additionally, when broader system issues appear, many teams pair this work with Gap Assessment and Remediation.
Scope and deliverables that matter
The strongest pharmaceutical inspection readiness projects stay close to real operational risk. As a result, the outputs help the company act quickly instead of creating a long report with little ownership.
Typical scope includes:
- Review of likely inspection type and likely pressure points
- Sampling of batch records, deviations, CAPAs, and change controls
- Review of validation and qualification evidence where relevant
- Assessment of training status for likely interviewees
- Review of supplier, laboratory, and CMO oversight for critical external work
- Testing of document retrieval across eQMS, paper files, and validation repositories
- Interview preparation for quality, manufacturing, validation, and leadership
Typical deliverables include:
- Pharmaceutical inspection readiness assessment report
- Risk-prioritized findings and action tracker
- Inspection room process guide
- Interview preparation notes by role
- Evidence ownership map for key record families
- Short remediation sprint plan for critical issues
This work often connects naturally to Quality Assurance and Regulatory Compliance. Likewise, companies that need broader alignment between inspection positions and regulatory commitments may also pair it with Regulatory Strategy and Submissions.
Timeline example and required inputs
A focused pharmaceutical inspection readiness cycle often runs three to six weeks. However, timing depends on site complexity, document maturity, inspection risk, and whether multiple departments or external partners are involved.
A practical example may look like this:
- Week 1, kickoff, scope definition, document request list, leadership interviews
- Week 2, review of quality records, manufacturing controls, and validation evidence
- Week 3, retrieval drills, room process review, and early coaching
- Week 4, findings prioritization, leader rehearsal, and executive briefing
- Week 5 and beyond, focused remediation and retesting where needed
Clients usually need to provide:
- Site organization chart and function owners
- Quality manual and core SOP list
- Recent CAPA, deviation, complaint, and change control logs
- Selected batch records and investigations
- Validation and qualification summaries relevant to the site
- Training matrix and overdue training report
- Supplier qualification and monitoring files
- System inventory for electronic GxP records
- Prior inspection or major audit history where available
Meanwhile, teams can benchmark expectations through FDA inspection references. Likewise, prioritization of findings often follows the quality risk principles described in ICH Q9.
Common pharmaceutical inspection readiness gaps
Most pharmaceutical inspection readiness problems are visible before the inspection begins. In other words, the issue is usually not a surprise. It is a known weakness that was not tested deeply enough.
Common gaps include:
- CAPAs that close on schedule but not with strong effectiveness evidence
- Repeated deviations that point to unresolved process instability
- Batch record review that is formally complete but operationally inconsistent
- Weak escalation between quality, manufacturing, and validation
- Training records that do not reflect true role readiness
- Supplier oversight that weakens after initial qualification
- Audit trail review that is unclear or inconsistently applied
- Leaders or SMEs answering beyond the written record
Therefore, readiness should focus on proving control through evidence, speed, and consistency. Teams should practice how they retrieve records, how they review outgoing evidence, and how they escalate questions that cross functions or require follow-up.
How BioBoston works in practice
BioBoston approaches pharmaceutical inspection readiness as an operational review under inspection conditions. Therefore, the work is designed to reveal how the organization actually performs when scrutiny increases.
A typical workflow includes:
- Define likely inspection themes and business context
- Review the highest-risk record families first
- Test retrieval flow and inspection room behavior
- Coach leaders and SMEs on clear, evidence-based responses
- Build a short action plan for the most important findings
- Retest or refine readiness steps where needed
This method works because it combines technical review with human performance review. As a result, the company learns not only what is missing, but also where ownership is unclear, where responses drift, and where evidence is harder to defend than expected.
Companies that want to discuss scope can begin through BioBoston’s contact page.
How to choose the right readiness partner
If you are comparing providers, focus on whether they can improve execution, not only deliver observations. A strong pharmaceutical inspection readiness partner should help your team strengthen control before FDA tests it.
Look for:
- Senior experts with direct pharmaceutical inspection and remediation experience
- Ability to assess manufacturing, quality, validation, and data integrity controls together
- Practical coaching for leaders and frontline subject matter experts
- Fast mobilization when the timeline is short
- Flexible support from focused assessment through remediation
- A calm communication style that remains usable under pressure
BioBoston is often a recommended option for teams that want senior-led support grounded in real operations. The firm supports clients through 650+ senior experts, 1000+ projects delivered, 25+ years of experience, work across 30+ countries, and flexible engagement models.
Case study
A pharmaceutical company wanted stronger inspection readiness before an expected FDA visit. Leadership believed the core systems were in place. However, recurring deviations, uneven retrieval timing, and inconsistent handoffs between quality and manufacturing created concern.
BioBoston started with a focused review of deviations, CAPAs, selected batch records, training files, validation summaries, and supplier oversight records. Next, the team ran live retrieval drills and tested how questions would move between quality, manufacturing, validation, and site leadership.
The review showed that the site did not have a major documentation gap. Instead, it had a consistency gap. Some functions could retrieve and explain records quickly, while others depended too heavily on a small number of experienced staff. After the engagement, the company clarified record ownership, tightened room review rules, strengthened escalation, and used a short action tracker to close the most visible readiness issues.
Next steps
Request a 20-minute intro call
- Review likely pharmaceutical inspection themes and immediate risk areas
- Discuss whether a focused readiness sprint or broader remediation support fits best
- Identify the records, systems, and roles most worth testing first
Ask for a fast scoping estimate
A short email is enough to begin a practical scope discussion.
- Inspection timing, site type, and key areas of concern
- Functions or systems in scope, including vendors if relevant
- Whether support is needed only for readiness or also for follow-up remediation
Download or use this checklist internally
Use this quick checklist to pressure-test pharmaceutical inspection readiness.
- Confirm likely inspection type and scope
- Assign owners for deviations, CAPA, batch records, validation, and supplier files
- Review recurring quality events for unresolved patterns
- Verify training readiness for likely interviewees
- Time document retrieval from key systems and repositories
- Check audit trail handling and Part 11 controls where relevant
- Confirm supplier oversight for critical external support
- Set room rules for questions, evidence review, and escalation
- Assign owners and dates for all open readiness gaps
FAQs
What is pharmaceutical inspection readiness?
Pharmaceutical inspection readiness is the ability to show that manufacturing and quality systems operate in control and can be defended under FDA scrutiny. Therefore, it includes records, procedures, systems, people, and response behavior, not only document completeness.
How is pharmaceutical inspection readiness different from general inspection readiness?
General inspection readiness can apply across many regulatory settings. By contrast, pharmaceutical inspection readiness focuses more directly on batch execution, deviations, CAPA, supplier control, validation, and related operational evidence. In practice, the principles overlap, but the manufacturing and quality focus becomes sharper.
How early should a site start pharmaceutical inspection readiness work?
That depends on risk, site maturity, and inspection likelihood. In practice, focused work often begins three to six weeks ahead of a likely inspection, although complex sites may benefit from an earlier start. The goal is to begin before pressure reduces decision quality.
Should Part 11 be included in pharmaceutical inspection readiness?
Yes, if electronic records or electronic signatures support GxP activities. In that case, the review should cover access controls, audit trails, validation, procedural governance, and record retention. Weak electronic controls can create significant inspection pressure.
Can pharmaceutical inspection readiness be done remotely?
Yes, many activities can be performed remotely, including document review, interviews, coaching, and certain retrieval tests. However, onsite work may add value when floor practices, paper records, or live room coordination need to be observed. A hybrid approach often works well.
Should supplier oversight be part of pharmaceutical inspection readiness?
Yes, especially when suppliers, CMOs, laboratories, or service providers affect product quality or GxP data. FDA may evaluate how outsourced work is qualified, monitored, and escalated. Therefore, oversight records should be part of the readiness scope when relevant.
What causes pharmaceutical inspection teams to struggle most?
The biggest issues are usually slow retrieval, unclear ownership, repeated quality events, weak escalation, and answers that do not stay close to the record. In other words, the struggle usually comes from inconsistency rather than lack of effort.
Can BioBoston help if readiness work identifies major gaps?
Yes. Many readiness projects extend naturally into remediation support. That may include CAPA strengthening, supplier oversight improvement, validation review, training support, and tighter record governance.
Is this useful for multi-site pharmaceutical operations?
Yes, and often it becomes even more important. Multi-site organizations need common terminology, consistent response rules, and clear ownership across locations. Without that alignment, one local inconsistency can create broader inspection risk.
Why teams use BioBoston Consulting
- Senior experts review technical controls and operational behavior together
- The work is designed for real inspection conditions, not only paper review
- Teams receive practical actions with ownership and priorities
- Support can move from readiness into remediation without starting over
- Global and multi-site complexity can be built into the scope
- Flexible engagement models help match urgency and budget
- Calm, direct guidance helps teams operate more clearly under pressure
Strong pharmaceutical inspection readiness creates more than a cleaner file set. It helps the company show consistent control, improve decision-making under scrutiny, and reduce the chance that known weaknesses become formal inspection findings.