
Best Pre-Approval Inspection Readiness: 7 Clear Steps
Pre-approval inspection readiness helps teams align filings, site controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

Pre-approval inspection readiness helps teams align filings, site controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate
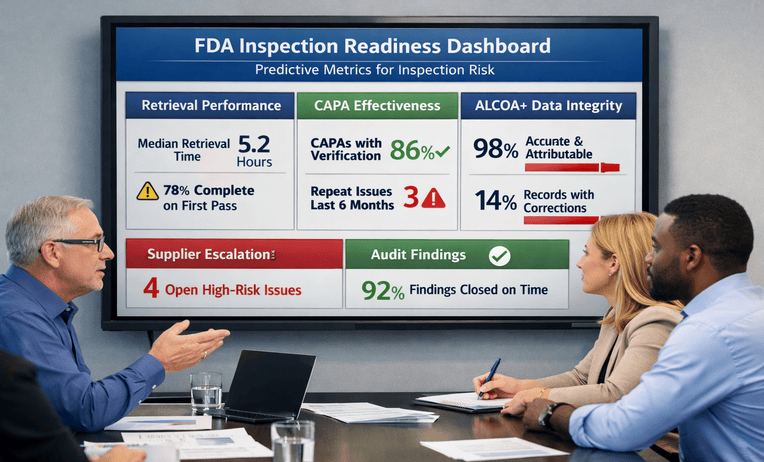
Pharmaceutical inspection readiness helps teams test records, controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

FDA inspection readiness training helps teams answer clearly, handle records, and escalate well under scrutiny. Build confidence and ask for a quick estimate

Quality system inspection readiness helps teams test CAPA, complaints, training, and oversight before scrutiny. Reduce risk and ask for a quick estimate

FDA audit preparation helps teams test records, roles, and systems before scrutiny begins. Reduce confusion and ask for a quick estimate

GMP inspection readiness helps manufacturing teams test records, systems, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

Mock FDA inspection helps teams test records, responses, and room behavior before scrutiny begins. Surface risks early and ask for a quick estimate

Inspection readiness checklist helps teams test records, systems, and roles before FDA scrutiny. Find gaps early and ask for a quick estimate

FDA 483 readiness helps teams prevent observations and respond clearly when issues arise. Build control before pressure increases. Ask for a quick estimate

FDA inspection readiness helps teams prepare records, people, and systems before scrutiny begins. Reduce risk and plan clearly. Request a scoping call

FDA inspection readiness for biotech companies helps teams align records, oversight, and responses before scrutiny. Reduce risk and ask for a quick estimate
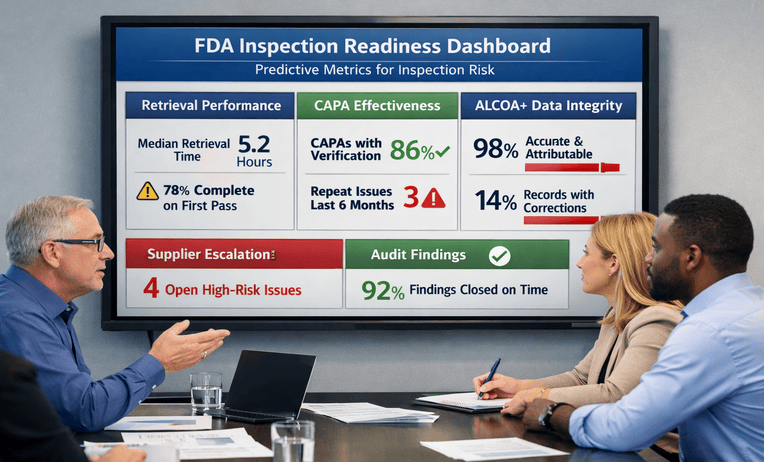
FDA inspection preparation services help teams test records, retrieval, and responses before scrutiny. Reduce risk and ask for a quick estimate