
Best Pre-Approval Inspection Readiness: 7 Clear Steps
Pre-approval inspection readiness helps teams align filings, site controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

Pre-approval inspection readiness helps teams align filings, site controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate
Pharmaceutical inspection readiness helps teams test records, controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

FDA inspection readiness training helps teams answer clearly, handle records, and escalate well under scrutiny. Build confidence and ask for a quick estimate

Quality system inspection readiness helps teams test CAPA, complaints, training, and oversight before scrutiny. Reduce risk and ask for a quick estimate

FDA audit preparation helps teams test records, roles, and systems before scrutiny begins. Reduce confusion and ask for a quick estimate

GMP inspection readiness helps manufacturing teams test records, systems, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

Mock FDA inspection helps teams test records, responses, and room behavior before scrutiny begins. Surface risks early and ask for a quick estimate

Inspection readiness checklist helps teams test records, systems, and roles before FDA scrutiny. Find gaps early and ask for a quick estimate

FDA 483 readiness helps teams prevent observations and respond clearly when issues arise. Build control before pressure increases. Ask for a quick estimate

FDA inspection readiness helps teams prepare records, people, and systems before scrutiny begins. Reduce risk and plan clearly. Request a scoping call
FDA inspection preparation services help teams test records, retrieval, and responses before scrutiny. Reduce risk and ask for a quick estimate

This article explores how life sciences organizations can systematically identify and remediate QMS gapshelping strengthen quality systems, reduce regulatory risk, and maintain a consistent state of inspection readiness.

Establishing an effective QMS requires more than implementation. It requires ongoing alignment, continuous improvement, and practical support that ensures the system works as intended under real operating conditions.

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

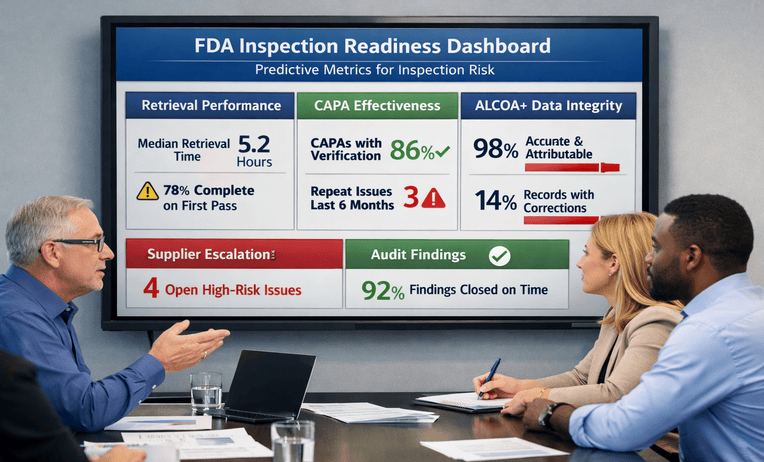
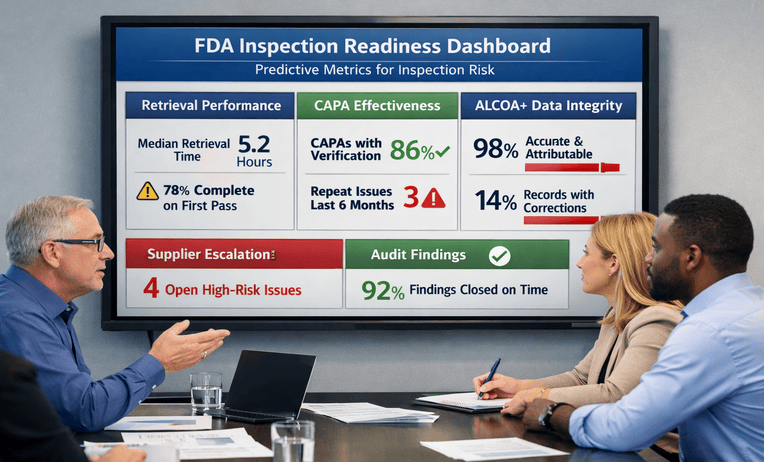
An FDA inspection readiness plan that strengthens CAPA, suppliers, and risk linkage with fast retrieval. Reduce surprises and build confidence. Ask for a quick estimate.

Remote FDA inspection readiness builds a reliable request workflow, controlled document room, and fast retrieval drills. Reduce chaos and stay calm. Ask for a quick estimate.

Pre-approval inspection readiness maps commitments to retrievable evidence across QMS, systems, and vendors. Reduce surprises and stay calm. Request a scoping call.

Form 483 response support turns observations into CAPA with verifiable effectiveness and fast retrieval. Reduce follow-up risk and regain control. Ask for a quick estimate.

First FDA inspection readiness keeps evidence retrievable across SOPs, data, and vendors. Reduce surprises and move faster. Request a scoping call.

FDA supplier audit readiness strengthens qualification, agreements, and follow-up so records are retrievable fast. Reduce supplier risk and pressure. Ask for a quick estimate.

FDA inspection readiness training built on timed drills improves retrieval speed and consistent answers across teams. Reduce surprises. Ask for a quick estimate.

Data integrity readiness strengthens ALCOA+ proof across systems, reviews, and vendors so records are retrievable fast. Reduce surprises. Request a scoping call.

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

If your inspection risk lives in systems and data, readiness looks different. Inspectors will ask how records are created, reviewed, corrected, and protected. Then they

Your inspection risk is not only in SOPs. It is in batch records, vendor oversight, data flows, and how quickly you can show evidence under

An FDA inspection can move fast, especially when inspectors ask for records across sites, systems, and vendors. As the QA leader, you are expected to

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

How to Develop an FDA-Inspection-Ready Quality Management System For biotech and pharmaceutical companies, regulatory inspections are not simply compliance events. They are a comprehensive evaluation

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because procedures are missing. More often, challenges arise when the Quality Management System (QMS)

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

QMS Inspection Readiness: Are You FDA-Prepared? An FDA inspection rarely fails because a company lacks procedures. It fails because the Quality Management System (QMS) does

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

Introduction: Ensuring Compliance and Quality Across Life Sciences In the pharmaceutical, biotechnology, and medical device industries, regulatory compliance is critical. Companies must not only meet

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

Introduction: Ensuring Compliance and Quality Across Life Sciences In the pharmaceutical, biotechnology, and medical device industries, regulatory compliance is critical. Companies must not only meet

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

What Is Quality Assurance and Regulatory Compliance? Quality Assurance (QA) and Regulatory Compliance are foundational pillars in the life sciences industry. QA ensures that products—from

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

For life sciences and biopharma companies, being prepared for an FDA inspection is essential to ensure GMP compliance and avoid regulatory setbacks. A structured FDA

For biopharma innovators, receiving an FDA Form 483 can be a significant regulatory challenge. Form 483 observations highlight potential GMP compliance issues identified during inspections,

In the life sciences industry, FDA inspections are not just regulatory milestones, they’re crucial moments that define your organization’s compliance, quality culture, and credibility. A

For biopharma innovators, receiving an FDA Form 483 can be a significant regulatory challenge. Form 483 observations highlight potential GMP compliance issues identified during inspections,

In the pharmaceutical industry, FDA inspections are crucial to ensure that manufacturing facilities, processes, and products comply with regulatory standards. Failing to meet FDA requirements

For life sciences and biopharma companies, being prepared for an FDA inspection is essential to ensure GMP compliance and avoid regulatory setbacks. A structured FDA

For life sciences and biopharma organizations, FDA inspections are critical milestones that test compliance with Good Manufacturing Practices (GMP). Without proper preparation, inspections can reveal

For pharmaceutical and biopharma companies, FDA Pre-Approval Inspections (PAIs) are critical steps in bringing new products to market. These inspections evaluate whether your manufacturing processes,

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

Ensuring Regulatory Compliance with FDA Inspection Readiness For Pharmaceutical, Biotechnology, and Medical device companies, an FDA inspection is a critical event that validates compliance with GxP and cGMP regulations. Being prepared for an FDA inspection

Proactive Compliance Across Global Regulatory Agencies For Pharmaceutical, Biotechnology, and Medical device companies, inspections from the FDA, EMA, and other global regulatory agencies are critical checkpoints that validate operational compliance, data integrity, and

Many of the clients we support come to us after experiencing inconsistent documentation, data-integrity findings, or challenging interactions with regulators. They often have strong science,

In many of the organizations we support, the first signs of compliance stress appear in fragmented documentation, incomplete audit trails, or inconsistencies across electronic systems.

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

Quality teams carry a heavy burden today. We often meet organizations where SOPs have grown in silos, documentation has lost consistency, and inspection readiness depends

Many life sciences organizations discover that quality issues surface long before an inspection. These issues such as gaps in documentation, inconsistent workflows, or unclear ownership

Imagine this scenario: A mid-size pharmaceutical manufacturer had been operating with what they believed was a “strong enough” quality system. Their last FDA inspection was several

Understanding FDA Inspections in the Biotech Industry For biotechnology and life sciences companies, FDA inspections are a critical component of regulatory compliance. These inspections evaluate a company’s adherence to GxP guidelines, cGMP

Passing internal audits does not always translate to inspection success. We often see Life Sciences organizations perform well on paper but struggle when FDA or

FDA inspections often focus attention on areas of highest regulatory and patient safety risk. We frequently see Life Sciences organizations allocate audit and readiness resources

Passing internal audits does not always translate to inspection success. We often see Life Sciences organizations perform well on paper but struggle when FDA or

Regulatory inspections today go far beyond verifying that procedures exist. We often see Life Sciences organizations pass internal audits yet face scrutiny during FDA or

For many Life Sciences organizations, FDA inspections are a source of significant stress. Teams may worry about unexpected questions, gaps in documentation, or supplier oversight,

As Life sciences organizations increasingly adopt cloud-based, SaaS, and agile technologies, ensuring regulatory compliance and system validation becomes more complex. We often see gaps in

The Life sciences and Biotech sectors are evolving rapidly, with organizations striving to bring innovative therapies, diagnostics, and medical devices to market efficiently and compliantly.

BioBoston Consulting | Audit-Driven QMS Readiness for Life Sciences Quality Management Systems are tested not only by regulators but also by partners, investors, and acquirers.

BioBoston Consulting | Inspection-Ready Audits and Proactive Regulatory Preparation FDA inspections can be high-stakes and stressful, particularly when teams are unsure how their systems, processes,

Most organizations believe they are inspection ready until inspectors start asking questions. We often see capable life sciences teams caught off guard by issues they did not expect, such as QMS

Imagine this scenario: A mid-size Pharmaceutical manufacturer had been operating with what they believed was a “strong enough” quality system. Their last FDA inspection was several years ago, and aside

Are You Confident Your Organization Could Withstand an FDA Inspection? Even well-prepared life sciences organizations can face surprises during FDA inspections such as: QMS or operational

Many teams across Biotech, Pharma, and MedTech tell us they rely on consulting partners to navigate pressure points in development, compliance, and commercialization. We often meet organizations that are

Many life sciences organizations discover that quality issues surface long before an inspection. These issues such as gaps in documentation, inconsistent workflows, or unclear ownership

One Stop Solution for Life Sciences Many life sciences organizations have ambitious plans such as new programs to launch, new markets to enter, and increasing

The 10 Market Leaders Mastering FDA Inspection-Readiness When we review inspection outcomes across the industry, a clear pattern appears: the most resilient organizations treat inspection-readiness

Quality teams carry a heavy burden today. We often meet organizations where SOPs have grown in silos, documentation has lost consistency, and inspection readiness depends

When we work with new clients, a familiar pattern often emerges. Teams are committed and capable, but their Quality Management System has not kept pace

In many of the organizations we support, the first signs of compliance stress appear in fragmented documentation, incomplete audit trails, or inconsistencies across electronic systems.

Many of the clients we support come to us after experiencing inconsistent documentation, data-integrity findings, or challenging interactions with regulators. They often have strong science,

Are You Truly Prepared for an FDA Inspection? Could gaps in your documentation or processes cause delays in FDA approval? Are your SOPs and quality

Shaping the Future of the Life Sciences Industry The global life sciences ecosystem is evolving faster than ever before. As breakthroughs emerge in biotechnology, pharmaceuticals,

In Life sciences, an FDA inspection is more than a regulatory milestone it is a defining moment. It reveals the strength of your systems, the maturity of your

Shaping the Future of the Life Sciences Industry The global life sciences sector is advancing at an unprecedented pace, as organizations work to transform breakthrough

Proactive Compliance Across Global Regulatory Agencies For Pharmaceutical, Biotechnology, and Medical device companies, inspections from the FDA, EMA, and other global regulatory agencies are critical checkpoints that validate operational compliance, data integrity, and

Ensuring Regulatory Compliance with FDA Inspection Readiness For Pharmaceutical, Biotechnology, and Medical device companies, an FDA inspection is a critical event that validates compliance with GxP and cGMP regulations. Being prepared for an FDA inspection

Simplifying Compliance in a Complex Regulatory Landscape In the highly regulated Life sciences industry, achieving FDA inspection readiness can feel overwhelming. Pharmaceutical, Biotech, and Medical device companies face increasing scrutiny from

Ensuring Regulatory Excellence in Life Sciences For Pharmaceutical, Biotechnology, and Medical device companies, FDA inspection readiness is not just about passing an audit, it is about demonstrating a strong culture of compliance, data integrity, and

Shaping the Future of the Life Sciences Industry The global life sciences landscape is advancing at a record pace, with organizations striving to convert scientific

Are You Truly Prepared for an FDA Inspection? Could gaps in your documentation or processes cause delays in FDA approval? Are your SOPs and quality

Ensuring Compliance for Regulatory Approvals The submission of new drug applications (NDAs) and biologic license applications (BLAs) is a critical step for Pharmaceutical and Biotech companies. Regulatory agencies, particularly

Ensuring Compliance in Clinical Trials In the Pharmaceutical and Biotechnology industries, clinical trial sponsors and Contract Research Organizations (CROs) are held to strict regulatory standards. Ensuring FDA inspection readiness is critical

Proactive Compliance in a GxP-Regulated World In today’s life sciences industry, compliance is more than a regulatory requirement; it is a foundation for patient safety, data integrity,

Building a Culture of Continuous Audit Readiness In the Life sciences industry, regulatory inspections are pivotal checkpoints that validate compliance, data integrity, and patient safety. Whether in clinical research or manufacturing operations, being inspection-ready is

Staying Ahead in a Regulated Industry In the Life sciences sector, regulatory audits are more than just checkpoints; they are critical milestones that reflect the integrity, quality, and reliability

In the Life sciences industry, FDA inspections are pivotal events that define your organization’s credibility, compliance, and operational maturity. FDA inspection readiness is more than just passing audits, it is about

Why FDA Inspection Readiness Is Essential for Pharma and Biotech Companies In the pharmaceutical and biotechnology industries, maintaining compliance with FDA regulations is critical to ensure product safety, efficacy, and market credibility. FDA inspections assess

Understanding FDA Inspections in the Biotech Industry For biotechnology and life sciences companies, FDA inspections are a critical component of regulatory compliance. These inspections evaluate a company’s adherence to GxP guidelines, cGMP

Understanding GxP and Its Role in FDA Compliance In the life sciences, pharmaceutical, and biotech industries, GxP compliance serves as the foundation for maintaining product quality, patient safety, and regulatory integrity. The

Strengthen Compliance, Minimize Risk, and Build Audit Confidence with BioBoston Consulting In the highly regulated world of Pharmaceuticals, Biotech, and Medical devices, achieving audit-ready operations is essential to maintain FDA compliance and ensure long term

In the Life sciences industry, FDA inspections are pivotal moments that define your organization’s credibility, compliance, and operational maturity. Being inspection ready is not just about passing audits, it is about

For life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations, from documentation and processes to staff readiness. Mock FDA audits

For pharmaceutical and biopharma companies, FDA Pre-Approval Inspections (PAIs) are critical steps in bringing new products to market. These inspections evaluate whether your manufacturing processes,

For life sciences and biopharma organizations, FDA inspections are critical milestones that test compliance with Good Manufacturing Practices (GMP). Without proper preparation, inspections can reveal

For life sciences and biopharma companies, being prepared for an FDA inspection is essential to ensure GMP compliance and avoid regulatory setbacks. A structured FDA

In the pharmaceutical industry, FDA inspections are crucial to ensure that manufacturing facilities, processes, and products comply with regulatory standards. Failing to meet FDA requirements

For biopharma innovators, receiving an FDA Form 483 can be a significant regulatory challenge. Form 483 observations highlight potential GMP compliance issues identified during inspections,

In the life sciences industry, FDA inspections are not just regulatory milestones—they’re crucial moments that define your organization’s compliance, quality culture, and credibility. A proactive

Master Every FDA Inspection with Confidence For biotech, pharmaceutical, and life sciences organizations, FDA inspections are high-stakes events that test every aspect of operations —

Navigating the Complexities of FDA Compliance For biotech, pharmaceutical, and life sciences organizations, achieving FDA inspection readiness is a complex, high-stakes process. From ensuring GxP

Ensuring Compliance, Confidence, and Readiness for Every FDA Inspection Preparing for an FDA inspection can be a defining moment for any biotech, pharmaceutical, or life

Future-Proof Your Organization for FDA Compliance In today’s rapidly evolving biotech and pharmaceutical landscape, FDA inspections demand more than reactive compliance. Organizations must align people,

Achieve Regulatory Confidence with BioBoston Consulting For biotech, pharmaceutical, and life sciences organizations, FDA inspections are pivotal events that test your operational compliance, documentation integrity,

Transforming Compliance from Event-Driven to Continuous Excellence For biotech, pharmaceutical, and life sciences organizations, FDA inspections can be high-pressure events. Waiting until an audit is

Preparing for an FDA inspection is a critical business moment for life sciences organizations. BioBoston Consulting delivers focused FDA Inspection Readiness services that combine technical

Your One Stop Solution for Life Sciences In the life sciences industry, audits are essential for ensuring regulatory compliance, product quality, and operational excellence. A

Your One Stop Solution for Life Sciences In the biopharma and biotech industries, ensuring 21 CFR Part 11 compliance in digital systems is essential for

Your One Stop Solution for Life Sciences In the biopharmaceutical and biotech industries, being prepared for internal and external audits is critical to maintaining regulatory

Your One Stop Solution for Life Sciences In the pharmaceutical and biotech industries, organizations often conduct frequent internal and supplier audits to maintain compliance and

One Stop Solution for Life Sciences 🔍 Why Data Integrity Is a Cornerstone of FDA Compliance In the life sciences industry, data integrity is not

One Stop Solution for Life Sciences 🔍 Demystifying FDA Inspection Types in Life Sciences For pharmaceutical, biotech, and medical device companies, FDA inspections are a

One Stop Solution for Life Sciences 🔍 What Is FDA Inspection Readiness and Why It Matters For pharmaceutical, biotech, and medical device companies, FDA inspection

One Stop Solution for Life Sciences 🌍 Why Global Manufacturers Must Prioritize FDA Inspection Readiness For international pharmaceutical, biotech, and medical device manufacturers exporting to

One Stop Solution for Life Sciences 🔍 Why FDA Inspection Readiness Is Non-Negotiable For pharmaceutical, biotech, and medical device manufacturers, being prepared for an FDA

One Stop Solution for Life Sciences 🔍 Why Mock FDA Inspections Are Critical for Compliance In the life sciences industry, FDA inspections are inevitable—and often

One Stop Solution for Life Sciences 🧬 What Is Quality Assurance and Regulatory Compliance? Quality Assurance (QA) and Regulatory Compliance are foundational pillars in the

One Stop Solution for Life Sciences 🏛️ What Is FDA Inspection Readiness? FDA Inspection Readiness is a proactive, strategic approach that prepares life sciences companies

In the biotech industry, few things disrupt operations and damage reputations like receiving an FDA Form 483. These inspectional observations highlight areas where a company

When the FDA arrives for an inspection, biotech companies must be ready to demonstrate compliance, transparency, and control. Success hinges not only on systems and

For biotech startups, securing FDA approval is a major milestone but passing an FDA inspection is often the first real test of operational maturity. With

One Stop Solution for Life Sciences ✅ What Is FDA Inspection Readiness? FDA Inspection Readiness refers to the strategic preparation that life sciences companies—pharmaceutical, biotech,

Introduction: Making Compliance Manageable In the life sciences industry, maintaining regulatory compliance and achieving FDA inspection readiness can be challenging. From pharmaceuticals to medical devices

Introduction: Preparing for FDA Inspections with Confidence FDA inspections are a critical component of regulatory oversight for pharmaceuticals, biologics, and medical devices. Being fully prepared

Introduction: Confidently Navigate FDA Inspections In the life sciences industry, FDA inspections are a critical component of regulatory oversight. Successfully managing these inspections ensures FDA

Introduction: Ensuring Compliance Across Every Level In the life sciences sector, achieving FDA inspection readiness requires more than just proper documentation—it demands a comprehensive approach

Introduction: Proactive Preparation is Key In the life sciences industry, FDA inspection readiness is critical to maintain compliance, protect product approvals, and ensure operational excellence.

Introduction: Staying Ahead in Life Sciences Compliance In the life sciences industry, unexpected FDA inspections can disrupt operations and delay product approvals. Proactive inspection readiness

Introduction: Ensuring Compliance in Life Sciences In the highly regulated life sciences sector, achieving FDA compliance and maintaining inspection readiness are critical for the successful

One Stop Solution for Life Sciences In life sciences—whether pharmaceuticals, biotech, medical devices, or diagnostics—maintaining product safety, compliance, and operational excellence depends strongly on your

One Stop Solution for Life Sciences In life sciences—pharmaceuticals, biotech, medical devices, diagnostics—FDA (Food and Drug Administration) inspections are central markers of regulatory compliance, product

Understanding Regulatory Compliance in Pharma and Biotech Navigating the life sciences industry requires strict adherence to regulatory requirements. Pharmaceutical, biotechnology, and medical device companies must

Why Harmonization Matters in Life Sciences In the pharmaceutical, biotechnology, and medical device sectors, companies operate across global markets. Navigating multiple regulatory frameworks—FDA, EMA, ICH,

Evolving CSV Practices in Life Sciences In the life sciences sector, Computer System Validation (CSV) is essential for ensuring compliance, patient safety, and product quality.

Why CSV Documentation Matters for FDA Inspections In today’s highly regulated life sciences industry, Computer System Validation (CSV) is not just a compliance requirement, it’s

Why Part 11 Compliance Matters for Life Sciences In the life sciences industry, electronic records and signatures are central to daily operations. The FDA’s 21

Why CSV Matters in Manufacturing In pharmaceutical, biotech, and medical device industries, Computer System Validation (CSV) is critical for ensuring compliance with FDA current Good

Why CSV Is Critical for Laboratory Systems In today’s life sciences environment, laboratories play a central role in quality control (QC) and research and development

Understanding Quality Management Systems in Life Sciences A Quality Management System (QMS) is the backbone of regulatory compliance for pharmaceutical, biotechnology, and medical device companies.

What Are Mock FDA Inspections? Mock FDA inspections are simulated regulatory audits designed to prepare pharmaceutical, biotechnology, and medical device companies for actual FDA inspections.

Why Team Training is Critical for FDA Inspection Readiness Preparing for an FDA inspection is not just about systems and documentation—it’s about people. A well-trained

Understanding FDA Inspection Readiness in Clinical Research In the highly regulated life sciences sector, FDA inspection readiness is essential for both sponsors and Contract Research

Understanding FDA 483 Observations When the U.S. Food and Drug Administration (FDA) inspects pharmaceutical, biotechnology, or medical device facilities, the inspector may issue a Form

Understanding Data Integrity in Life Sciences Data integrity is a critical component of FDA compliance in the pharmaceutical, biotechnology, and medical device industries. It ensures

One Stop Solution for Life Sciences What Are Internal and Supplier Audits? In the life sciences industry—pharmaceuticals, biotechnology, and medical devices—audits are a cornerstone of

Introduction: Internal Audits Beyond Compliance In the life sciences sector covering pharmaceuticals, biotechnology, and medical devices, internal audits are often seen as a compliance requirement.

Introduction: Audits as a Cornerstone of Compliance For pharmaceutical, biotechnology, and medical device companies, inspection readiness is not optional—it’s essential. Regulatory bodies such as the

Introduction: Ensuring Compliance and Quality Across Life Sciences In the pharmaceutical, biotechnology, and medical device industries, regulatory compliance is critical. Companies must not only meet

Introduction: Turning Inspections Into Opportunities For pharmaceutical, biotechnology, and medical device organizations, FDA inspections are critical milestones that determine compliance, product approval, and market access.

Introduction: Why FDA Inspection Readiness Matters For pharmaceutical, biotechnology, and medical device companies, FDA inspections are not just regulatory requirements—they are critical checkpoints that determine

Introduction: Turning FDA Compliance Into an Advantage In the life sciences industry, FDA readiness is more than a regulatory requirement, it is a strategic advantage.

Introduction: Why Inspection Readiness Is Essential In today’s highly regulated environment, pharmaceutical and biotech companies cannot afford to take FDA inspections lightly. Being inspection-ready is

Introduction: Taking the Fear Out of FDA Audits For pharmaceutical, biotechnology, and medical device companies, FDA audits are often viewed with apprehension. A single inspection

Introduction: Shifting From Fear to Confidence For many pharmaceutical, biotechnology, and medical device companies, the phrase FDA audit brings stress and uncertainty. But inspections don’t

One Stop Solution for Life Sciences In the stringent regulatory landscape of life sciences—whether pharmaceuticals, biologics, or medical devices—being prepared for an FDA inspection is

In today’s competitive life sciences industry, success is defined not only by innovation but also by quality and compliance. For organizations developing pharmaceuticals, biologics, or

In the life sciences industry, compliance, quality, and patient safety are non-negotiable. One of the most effective tools organizations can use to safeguard their operations

In the life sciences industry, few events are as critical as an FDA inspection. Whether your company is developing pharmaceuticals, biologics, or medical devices, regulatory

In the life sciences industry, maintaining accurate and comprehensive quality assurance (QA) documentation and records is essential for regulatory compliance, operational efficiency, and product safety.

In the highly regulated life sciences industry, maintaining Quality Assurance (QA) excellence is essential for ensuring product quality, patient safety, and regulatory compliance. Companies that

For pharmaceutical, biotechnology, and medical device companies, FDA inspections are a critical component of regulatory compliance. Being prepared for these inspections not only demonstrates adherence

In the highly regulated life sciences industry, a robust Quality Management System (QMS) is critical for achieving compliance, operational efficiency, and successful product development. From

In the highly regulated life sciences industry, achieving and maintaining compliance with FDA regulations and global standards is non-negotiable. Whether operating under Good Manufacturing Practices

In the life sciences industry, suppliers play a critical role in ensuring the safety, quality, and compliance of products. From raw materials to critical components,

In the life sciences industry, regulatory inspections are inevitable. The FDA expects organizations to maintain high standards of compliance across GMP, GCP, and GLP environments.

In the highly regulated life sciences sector, GxP compliance covering GMP (Good Manufacturing Practice), GCP (Good Clinical Practice), and GLP (Good Laboratory Practice) is essential

In the highly regulated life sciences industry, compliance is not a one-time effort—it requires consistent oversight and continuous improvement. One of the most effective tools

In the life sciences industry, achieving and maintaining FDA inspection readiness is critical for success. Whether your organization is involved in Good Manufacturing Practices (GMP),

In the highly regulated life sciences industry, companies must consistently meet FDA standards across Good Manufacturing Practices (GMP), Good Clinical Practices (GCP), and Good Laboratory

In the competitive and highly regulated life sciences industry, regulatory compliance is non-negotiable. Companies operating under Good Manufacturing Practices (GMP), Good Clinical Practices (GCP), and

In the highly regulated life sciences industry, receiving an FDA Form 483 can be costly and damaging for any organization. These observations are issued when

In the life sciences sector, FDA inspections are a critical part of ensuring regulatory compliance and safeguarding patient safety. Organizations operating in Good Manufacturing Practices

In the highly regulated life sciences industry, FDA inspections are a critical checkpoint for ensuring compliance, product quality, and patient safety. Companies operating under Good

In the highly regulated life sciences industry, FDA inspections are a critical part of ensuring compliance, product quality, and patient safety. However, many companies face

One Stop Solution for Life Sciences In the highly regulated life sciences industry, preparing for an FDA inspection is crucial. BioBoston Consulting offers comprehensive FDA