Insights
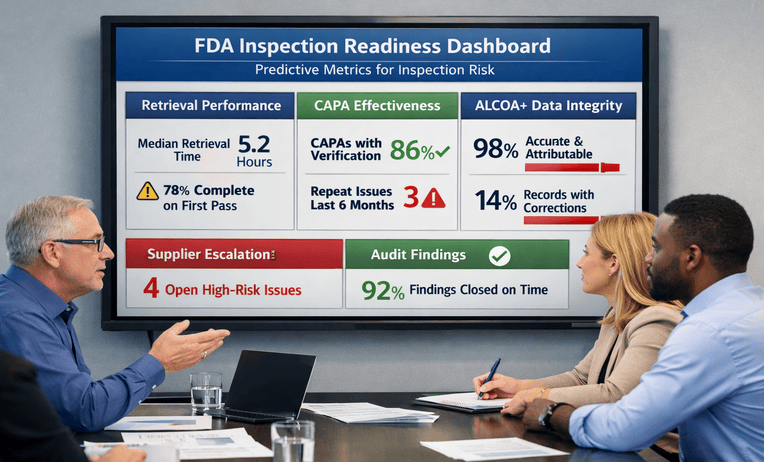
Best FDA Inspection Preparation Services: 8 Trusted Steps
FDA inspection preparation services help teams test records, retrieval, and responses before scrutiny. Reduce risk and ask for a quick estimate

9 Signs of Reliable IDE Submission Support for Device Studies
IDE submission support helps sponsors align risk, protocol, and study controls before filing. Request a scoping call today.

7 Smart Reasons to Choose the Right IDE Filing Support Partner
IDE filing support helps sponsors align risk, protocol, and study controls before submission. Request a scoping call today.

7 Clear Ways to Choose Best IDE Application Consulting Partner
IDE application consulting helps device teams build defensible submissions, reduce risk, and stay inspection ready. Ask for a quick estimate.

6 Factors Behind High-Quality IDE Submission Support
IDE submission support helps device sponsors align strategy, protocol, and compliance controls before filing. Ask for a quick estimate.

6 Smart Reasons to Choose the Right IDE Submission Consulting Support
IDE submission consulting support helps sponsors align risk, protocol, and study controls before filing. Ask for a quick estimate.
