Insights

10 Proven Traits of an Effective BLA Project Management Partner
BLA project management for biologics teams needing stronger filing control, clearer dependencies, and calmer execution. Request a scoping call.

How to Identify the Best BLA Readiness Partner for Biologics Companies: 10 Key Indicators
BLA submission readiness for biologics teams needing clearer filing maturity, stronger prioritization, and calmer decisions. Ask for a quick estimate.

10 Essential Qualities of a Trusted Biologics Submission Strategy Partner
Biologics submission strategy for teams that need a clearer BLA path, stronger readiness logic, and calmer execution. Request a scoping call.

10 Ways to Identify the Right BLA Readiness Review Partner
BLA readiness review for biologics teams that need clearer risk visibility, stronger governance, and calmer late-stage execution. Ask for a quick estimate.

Pre-Approval Inspection Readiness: 7 Critical Steps for Audit and Approval Readiness
Pre-approval inspection readiness helps teams align filings, site controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate
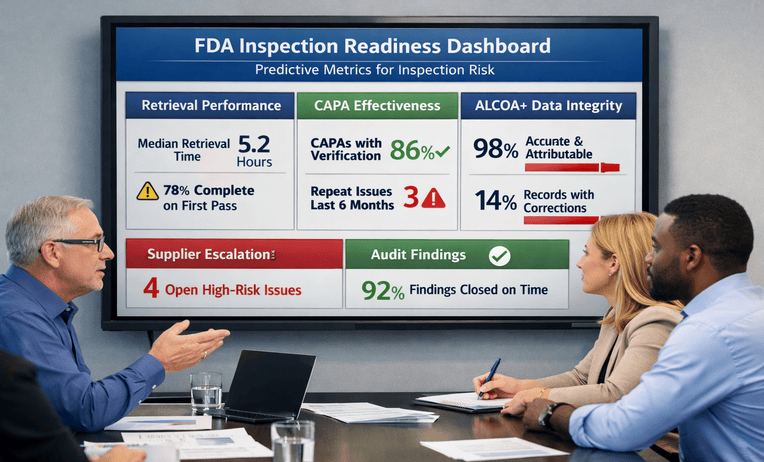
Pharmaceutical Inspection Readiness: 8 Strategic Steps for Audit and Compliance Success
Pharmaceutical inspection readiness helps teams test records, controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate
