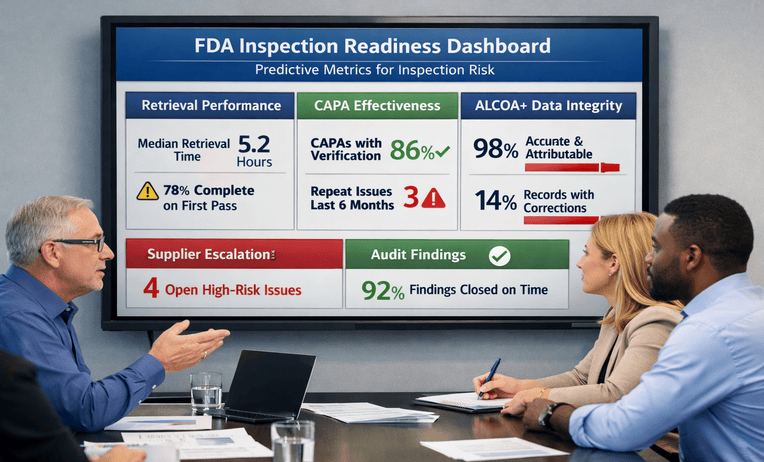
Regulatory inspections increasingly probe beyond documented procedures to evaluate how quality systems function in practice. We often see Life Sciences organizations pass internal audits yet struggle during FDA or global health authority inspections when inspectors follow data flows, supplier dependencies, and cross-functional decision-making. In many cases, the issue is not the absence of audits, but whether those audits truly reflect regulatory expectations.
At BioBoston Consulting, we design and execute internal and supplier audits built to withstand FDA and global health authority scrutiny. Our audit approach is grounded in real inspection behavior, helping organizations validate readiness, identify risk early, and strengthen oversight across GxP-regulated operations.
Why many audit programs fall short during inspections
Across Pharma, Biotech, and MedTech environments, we frequently observe that:
- Internal audits focus on SOP compliance rather than process effectiveness
- Supplier audits rely heavily on questionnaires and certifications
- Audit scopes do not reflect product risk, lifecycle stage, or inspection history
- Audit findings are documented but not clearly linked to regulatory impact
These gaps often surface during inspections, when regulators challenge how decisions are made, how data is controlled, and how supplier quality issues are managed.
Audit programs aligned with regulatory inspection reality
BioBoston Consulting structures audits to mirror how FDA and global health authorities assess compliance. Our internal and supplier audits are risk-based, process-driven, and inspection-focused, ensuring they hold up under regulatory review.
Our audit services typically include:
- Risk-based internal audits targeting critical GxP processes, systems, and controls
- Supplier audits focused on material impact, data integrity, and quality agreement execution
- Data integrity audits aligned with ALCOA+ principles and global regulatory guidance
- End-to-end process audits, following records, deviations, and approvals across functions
- Inspection-ready audit reporting that clearly links observations to regulatory expectations
This approach helps organizations move beyond checklist audits toward meaningful risk assessment.
Strengthening global compliance and supplier oversight
Global health authorities increasingly expect consistent quality oversight across sites and third-party partners. We help organizations evaluate whether internal audit programs and supplier audits are aligned globally and supported by effective governance, escalation, and CAPA management.
Our audits assess:
- Oversight of contract manufacturers, labs, and critical vendors
- Effectiveness of quality agreements and performance monitoring
- Integration between internal audits, supplier audits, and management review
- Readiness for FDA, EMA, MHRA, and other global inspections
Practical outcomes that support inspection success
BioBoston Consulting audits are designed to deliver clarity and action. Clients receive:
- Risk-ranked audit observations with regulatory context
- Clear insight into inspection exposure and priority gaps
- Support for CAPA planning and remediation strategies
- Stronger alignment between audit programs and inspection readiness efforts
If your internal and supplier audits were to be tested under FDA or global health authority scrutiny tomorrow, would they hold up? BioBoston Consulting can support risk-based internal and supplier audits that strengthen compliance and help organizations face inspections with confidence. Let us discuss how we can help prepare your audit program for regulatory reality.