
Best Biologics License Application Services: 7 Defensible Selection Criteria
Biologics license application services that help teams plan, align, and de-risk BLA execution with clear senior support. Ask for a quick estimate

Biologics license application services that help teams plan, align, and de-risk BLA execution with clear senior support. Ask for a quick estimate

Biologics regulatory strategy for teams that need a clearer BLA path, stronger readiness decisions, and calmer execution. Request a scoping call.

BLA submission support for emerging biotech teams that need clearer readiness, stronger review control, and calmer execution. Ask for a quick estimate.

BLA application services for biologics teams needing a clearer filing path, stronger readiness, and calmer execution. Request a scoping call.

BLA application services that bring structure, quality, and inspection-focused planning to biologics filings. Ask for a quick estimate

BLA gap assessment for biologics teams that need clearer risk visibility, stronger prioritization, and calmer filing decisions. Ask for a quick estimate.

Biologics filing governance support for teams that need clearer decision control, stronger escalation, and calmer BLA execution. Request a scoping call.

Biologics license application planning for teams that need stronger filing control, clearer dependencies, and calmer execution. Request a scoping call.

FDA biologics approval support for BLA teams needing clearer readiness, stronger filing control, and calmer execution. Request a scoping call.

BLA project management for biologics teams needing stronger filing control, clearer dependencies, and calmer execution. Request a scoping call.

Biologics submission strategy for teams that need a clearer BLA path, stronger readiness logic, and calmer execution. Request a scoping call.

BLA submission readiness for biologics teams needing clearer filing maturity, stronger prioritization, and calmer decisions. Ask for a quick estimate.

BLA readiness review for biologics teams that need clearer risk visibility, stronger governance, and calmer late-stage execution. Ask for a quick estimate.

Pre-approval inspection readiness helps teams align filings, site controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate
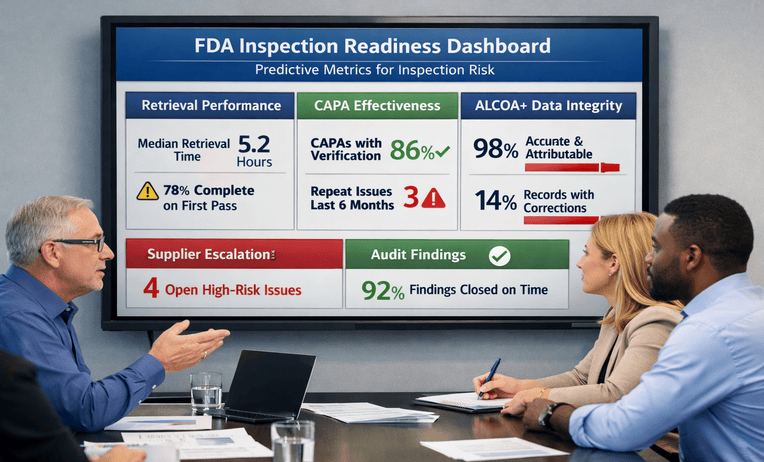
Pharmaceutical inspection readiness helps teams test records, controls, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

FDA inspection readiness training helps teams answer clearly, handle records, and escalate well under scrutiny. Build confidence and ask for a quick estimate

Quality system inspection readiness helps teams test CAPA, complaints, training, and oversight before scrutiny. Reduce risk and ask for a quick estimate

FDA audit preparation helps teams test records, roles, and systems before scrutiny begins. Reduce confusion and ask for a quick estimate

GMP inspection readiness helps manufacturing teams test records, systems, and responses before FDA scrutiny. Reduce risk and ask for a quick estimate

Mock FDA inspection helps teams test records, responses, and room behavior before scrutiny begins. Surface risks early and ask for a quick estimate